DocumentoHCF-Guidelines-Proposal Template-2021cargado por
DocumentoHCF-Guidelines-Proposal Template-2021cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMedical Devices Violation - Ver 2.1cargado por
DocumentoMedical Devices Violation - Ver 2.1cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoResolution No. (24) for 2021cargado por
DocumentoResolution No. (24) for 2021cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMedical Devices Authorized Representatives Check Listcargado por
DocumentoMedical Devices Authorized Representatives Check Listcargado por Mohamed Ezzat
Mohamed Ezzat DocumentoInspection Request Form (1)cargado por
DocumentoInspection Request Form (1)cargado por Mohamed Ezzat
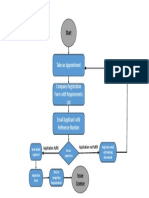
Mohamed Ezzat DocumentoProcess of Authorized Representative Registration (1)cargado por
DocumentoProcess of Authorized Representative Registration (1)cargado por Mohamed Ezzat
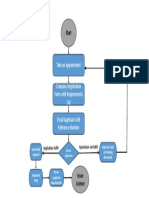
Mohamed Ezzat Documentoprocess of Authorized Representative registration (1)cargado por
Documentoprocess of Authorized Representative registration (1)cargado por Mohamed Ezzat
Mohamed Ezzat Documentoabc flayercargado por
Documentoabc flayercargado por Mohamed Ezzat
Mohamed Ezzat DocumentoPricing Proposal NHRAcargado por
DocumentoPricing Proposal NHRAcargado por Mohamed Ezzat
Mohamed Ezzat Documentoabc flayer_Modcargado por
Documentoabc flayer_Modcargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMagedSh - CVcargado por
DocumentoMagedSh - CVcargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMD Companies Registrationcargado por
DocumentoMD Companies Registrationcargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMagedSh - CL (1)cargado por
DocumentoMagedSh - CL (1)cargado por Mohamed Ezzat
Mohamed Ezzat Documentoproposal ar and mdrcargado por
Documentoproposal ar and mdrcargado por Mohamed Ezzat
Mohamed Ezzat DocumentoABC prop p1cargado por
DocumentoABC prop p1cargado por Mohamed Ezzat
Mohamed Ezzat Documentotable 2cargado por
Documentotable 2cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDD_11163-2017-CE-KOR-NA-PS Rev. 6.0cargado por
DocumentoMDD_11163-2017-CE-KOR-NA-PS Rev. 6.0cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoTwins and Siblingscargado por
DocumentoTwins and Siblingscargado por Mohamed Ezzat
Mohamed Ezzat DocumentoUzinmedicarecargado por
DocumentoUzinmedicarecargado por Mohamed Ezzat
Mohamed Ezzat DocumentoISO13485_10317-2017-AQ-KOR-NA-PS Rev. 2.0.- 20210413cargado por
DocumentoISO13485_10317-2017-AQ-KOR-NA-PS Rev. 2.0.- 20210413cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular 11_Prohibition of Importation of Transvaginal Mesh_2018cargado por
DocumentoMDR_Circular 11_Prohibition of Importation of Transvaginal Mesh_2018cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoCircular 12 2018cargado por
DocumentoCircular 12 2018cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular_21_2020_Extension of Temporary Holdup of Receiving New Medical Devices and Supplies Registration Requestscargado por
DocumentoMDR_Circular_21_2020_Extension of Temporary Holdup of Receiving New Medical Devices and Supplies Registration Requestscargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular 8_Classification of criteria of Combined Medical Device_2018cargado por
DocumentoMDR_Circular 8_Classification of criteria of Combined Medical Device_2018cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular_21_2020_Extension of temporary holdup of receiving new medical devices and supplies registration requestscargado por
DocumentoMDR_Circular_21_2020_Extension of temporary holdup of receiving new medical devices and supplies registration requestscargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular 8_2017cargado por
DocumentoMDR_Circular 8_2017cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular 7 _New Appointment Booking System_2019cargado por
DocumentoMDR_Circular 7 _New Appointment Booking System_2019cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular 10_Prohibition of Importation of Powdered Medical Gloves_2018cargado por
DocumentoMDR_Circular 10_Prohibition of Importation of Powdered Medical Gloves_2018cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular_17_2020_Guideline of hand sanitizerscargado por
DocumentoMDR_Circular_17_2020_Guideline of hand sanitizerscargado por Mohamed Ezzat
Mohamed Ezzat Documentono 45 for 2020cargado por
Documentono 45 for 2020cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular 14_New Details to clear Medical Devices Shipments_2017cargado por
DocumentoMDR_Circular 14_New Details to clear Medical Devices Shipments_2017cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular 5_Medical Devices Certificate Verification and Validation_2018cargado por
DocumentoMDR_Circular 5_Medical Devices Certificate Verification and Validation_2018cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular_7_2020_Rapid test for research purposes only and not for diagnosiscargado por
DocumentoMDR_Circular_7_2020_Rapid test for research purposes only and not for diagnosiscargado por Mohamed Ezzat
Mohamed Ezzat Documento2021_Circular No. (4) 2021cargado por
Documento2021_Circular No. (4) 2021cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoCircular No. ) 2 ( 2021cargado por
DocumentoCircular No. ) 2 ( 2021cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular 10_A new Document to clear the shipment of medical device_2017cargado por
DocumentoMDR_Circular 10_A new Document to clear the shipment of medical device_2017cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoMDR_Circular_6_2020_Air Doctor Productcargado por
DocumentoMDR_Circular_6_2020_Air Doctor Productcargado por Mohamed Ezzat
Mohamed Ezzat DocumentoNHRA Closed due to Shifting to New Locationcargado por
DocumentoNHRA Closed due to Shifting to New Locationcargado por Mohamed Ezzat
Mohamed Ezzat Documento401474910 CGP CGP GSCE Chemistry AQA Revision Guide 2018 PDFcargado por
Documento401474910 CGP CGP GSCE Chemistry AQA Revision Guide 2018 PDFcargado por Mohamed Ezzat
Mohamed Ezzat DocumentoWPI_Log_2017.03.26_17.33.58cargado por
DocumentoWPI_Log_2017.03.26_17.33.58cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoWPI_Log_2017.03.26_17.17.44cargado por
DocumentoWPI_Log_2017.03.26_17.17.44cargado por Mohamed Ezzat
Mohamed Ezzat Documentomktg-1030-swot-presentationcargado por
Documentomktg-1030-swot-presentationcargado por Mohamed Ezzat
Mohamed Ezzat DocumentoIndicescargado por
DocumentoIndicescargado por Mohamed Ezzat
Mohamed Ezzat DocumentoField Safety Noticecargado por
DocumentoField Safety Noticecargado por Mohamed Ezzat
Mohamed Ezzat Documentoreview report for renewcargado por
Documentoreview report for renewcargado por Mohamed Ezzat
Mohamed Ezzat DocumentoSupplementary Exercise 3 - Summary statisticscargado por
DocumentoSupplementary Exercise 3 - Summary statisticscargado por Mohamed Ezzat
Mohamed Ezzat DocumentoAdverse Event Procedurecargado por
DocumentoAdverse Event Procedurecargado por Mohamed Ezzat
Mohamed Ezzat DocumentoAdverse Event-procedure (2)cargado por
DocumentoAdverse Event-procedure (2)cargado por Mohamed Ezzat
Mohamed Ezzat DocumentoComplaint Formcargado por
DocumentoComplaint Formcargado por Mohamed Ezzat
Mohamed Ezzat
Documentos de Académico
Documentos de Profesional
Documentos de Cultura