Documentos de Académico
Documentos de Profesional
Documentos de Cultura
GDPMD Guide - Update 1st August 2014 Sheet1
Cargado por
naravichandran3662Título original
Derechos de autor
Formatos disponibles
Compartir este documento
Compartir o incrustar documentos
¿Le pareció útil este documento?
¿Este contenido es inapropiado?
Denunciar este documentoCopyright:
Formatos disponibles
GDPMD Guide - Update 1st August 2014 Sheet1
Cargado por
naravichandran3662Copyright:
Formatos disponibles
UPDATE:
August 1st 2014
GUIDE
FOR
THE
IMPLEMENTATION
OF
GOOD
DISTRIBUTION
PRACTICES
FOR
MEDICAL
DEVICE
(GDPMD)
Provision
no.
2
3
4
10
11
12
13
Provision
Requirements
Recommendations/Notes
Scope and application
GDPMD
is
applicable
to
Authorized
representatives
of
foreign
manufacturers,
importers
and
distributors,
IT
IS
Manufacturers
will
be
required
to
be
ISO
13485
certified.
NOT
APPLICAPLE
to
manufacturers
and
retailers
of
medical
devices.
Justification
has
to
be
provided
if
a
GDPMD
requirement
is
not
applicable
It
is
recommended
to
document
these
justification
in
the
Quality
Manual
or
Regulatory
Compliance
manual
GDPMD
certification
to
be
conducted
by
an
approved
Conformity
Assessment
Body
(CAB)
A
list
of
approved
CAB's
is
available
here:
http://www.mdb.gov.my/mdb/index.php?option=com_content&task=view&id
=185&Itemid=138
Definitions
"Authority"
means
the
Medical
Device
Authority
(MDA)
MDA
is
a
division
in
the
Ministry
of
Health
Malaysia
(MOH)
in
charge
of
regulating
medical
device
and
its
industry
players
in
the
country.
Organization
Define
the
organization
structure,
duties
and
responsibilities
Organizational
chart
with
names
and
positions
showing
interrelationship
of
all
key
personnel.
For
each
position,
develop
job
descriptions
with
title,
authorities
and
responsibilities.
Setup
AND
maintain
GDPMD
regulatory
compliance
system
It
is
stated
here
that
the
organization
shall
maintain
the
effectiveness
of
the
regulatory
compliance
system
by
implementing
appropriate
CAPA
system
GDPMD
Regulatory
Compliance
Establish,
document
and
implement
GDPMD
RCS
System
is
generally
described
in
the
Regulatory
Compliance
Manual
System
(RCS)
-General
Identify
proccesses
needed
for
the
GDPMD
Define
a
3
layers
process
map
showing
interaction:
management,
realization
and
support
Determine
criteria
and
methods
needed
for
the
control
of
these
processes
process
matrix
with
process
name,
pilot,
activities,
indicators,
review
frequency
and
effectiveness
criteria
(targets)
Ensure
resources
availability
for
each
process,
resources
needs
shall
be
reviewed
on
a
regular
basis
(during
management
reviews,
meetings,etc)
Monitor,
measure
and
analyze
these
processes
analyze
process
matrix
indicators
on
a
regular
basis
(it
is
a
good
practice
to
review
quarterly)
Implement
actions
to
achieve
planned
results
and
maintain
effectiveness
implement
CAPA
system
Identify
and
control
outsourced
processes
ie:
sterilization,
packaging...
a
close
monitoring
of
these
processes
is
also
required.
Documentation
Regulatory
Compliance
manual
equivalent
to
"Quality
Manual"
for
ISO13485.
Describe
clearly
which
medical
devices
the
establishment
is
dealing
with
and
their
status
of
compliance
(in
Malaysia
and
abroad)
make
a
reference
to
GDPMD
RCS
procedures,
other
documents/records
needed
for
compliance.
scope
of
GDPMD
RCS
including
details
of
and
justification
fo
any
exclusion
and/or
non-application
Specific
information
related
to
premises,
personnel
and
medical
device
conformity
assessment/registration
holder
Detailed
description
on
how
the
relevant
and
applicable
regulatory
requirements
are
adressed
for
each
Suggestion
here
is
to
prepare
a
checklist
with
requirements,
applicability
and
medical
device
specified
in
the
scipe
of
the
GDPMD
RCS
reference
documents
provided
as
a
compliance
objective
evidence
For
each
type
of
MD,
the
establishment
shall
maintain
a
file.
Similar
to
DMR
(Device
Master
Record)
for
a
manufacturer,
this
should
include
all
the
specifications
required
for
proper
handling
of
a
device
by
the
establishment
(it
is
device
specific)..
Document
control
Document
control
procedure
establish
a
procedure
on
how
to
control
documents
(incl.
numbering,
structure,
archiving,
disposition,
etc)
prepared,
approved,
signed
by
an
authorized
person
establish
a
signature
matrix
including
levels
of
authorization
for
each
type
of
document
prevent
unintended
use
of
superseded
version
include
change
control
in
document
control
procedure
establish
and
maintain
records
of
GDPMD
RCS
establish
an
instruction
for
Good
documentation
practices
define
controls
for
the
identification,
storage,
protection,
retrieval,
retention
time
and
disposition
establish
a
procedure
for
control
of
records
(incl.
list
of
GDPMD
records)
records
retention
determine
retention
time
for
each
type
of
record
Responsibilities
and
authorities responsibilities
and
authorities
to
be
documented
and
communicated
it
is
a
good
practice
to
have
responsibilities
and
authorities
documented
in
a
specific
section
for
each
procedure
interrelations
between
all
personnel
Organization
chart
(including
handling
personnel)
Designated
person
Appoint
designated
person
Equivalent
to
Management
representative.
This
is
generally
the
person
having
a
good
knowledge
of
GDPMD
requirements
(Quality
Manager,
Quality
Engineer,
QA
executive,
etc)
Suggestion:
a
letter
of
appoinment
should
be
issued
to
the
DP.
Management
review
Management
review
A
review
of
the
GDPMD
RCS
shall
be
organized
on
a
regular
basis
(ie.quarterly)
where
the
management
team
will
review
the
whole
system
to
ensure
compliance.
Minutes
of
the
management
review
shall
be
recorded.
Review
Input
results
of
internal
and
external
audit
management
review
shall
include
all
of
the
review
input
Review Output
Personnel
14
Training
15
Infrastrucure
16
Work environment
17
Cleanliness and pest control
18
Authorization
19
Communication channel
customer
complaint/feedback
GDPMD
regulatory
compliance
system
and
medical
device
compliance
surveillance
and
vigilancee
activities
including
field
safety
corrective
actions,
advisory
notes,
recalls
and
adverse
event/incident
reporting
feedback
from
manufacturer
feedback
and
directives
from
the
authority
status
of
preventive
and
corrective
actions
follow-up
actions
from
previous
management
review
changes
that
could
affect
the
GDPMD
recommendatios
for
compliance
corrective
and
preventive
actions
required
effectiveness
of
GDPMD
and
its
compliance
with
Malaysian
medical
device
regulatory
requirements
resource
needs
Personnel
shall
be
competent
and
possesses
appropriate
professional
knowledge,education,
training,
skills
and
experience.
Personnel
providing
post
market
technical
support
for
active
devices
shall
conform
to
the
requirements
and/or
standards
recognized
by
the
authority
Adequate
number
of
competent
personnel
involved
in
the
supply
chain
to
ensure
quality,
safety
and
performance
Determine
the
necessary
competences
Provide
training
Evaluate
training
effectiveness
Maintain
training
records,
skills
and
experience
Ensure
premises
ad
equipment
used
are
suitable,
secure,
safe
and
adequate
Establish
documented
requirements
management
review
shall
include
any
decisions
and
actions
related
to
the
review
output
Employee
Individual
file
shall
include
CV,
certificates,etc...
Technicians performing after sales service shall be trained to execute
maintenance activities as per manufacturer specifications and local regulations.
Adequate means enough resources to run the operations safely and efficiently.
This will depend on the company business requirements.
eg: during annual performance reviews
Suggestion: develop training needs based on performance review outcome
Training evaluation form with effectiveness evaluation section
Training records shall be part of the GDPMD system
Proper space for storage and handling of the product
Suggestion: Establish work instruction for maintaning the premises and
equipment, including their frequencies
Maintain records of maintenance activities
Records shall be part of the GDPMD system
Determine and manage work environment needed
eg. Special work environment such as controlled room needed?
Establish documented requirements for health, cleanliness and clothing of personnel if contact between such Suggestion: Establish work instruction for health, cleanliness and dressing of
personnel and medical devices or work environment that could adversely affect quality of medical devices
personnel
Establish documented procedure or work instructions to monitoring and control conditions for work
Establish procedure/work instruction for work environment monitoring
environment that could adversely affect quality of medical devices
(Temperature, Humidity)
Ensure all personnel who are required to work temporarily under special environment that could adversely This requirement can be included in procedure/work instruction for work
affect quality of medical devices are appropriately trained or supervised by a trained person
environment monitoring
Establish special arrangements and document the control of contaminated or potentially contaminated
This requirement can be included in procedure/work instruction for work
medical devices, work environment or personnel
environment monitoring
Establish documented requirements
Suggestion: Establish procedure/work instruction for cleaning of premises, pest
control program, including frequency and methods
Maintain records of cleaning
Cleaning records shall be part of the GDPMD system
Establish a pest control program
Suggestion: Establish procedure/work instruction for pest control program.
Pest control program can be managed internally or by pest control service
provider (eg. Installation of insect light trap (for flying insect) and installation of
rodent trap)
Maintain records of pest control program
Pest control records shall be part of the GDPMD system
Obtain required authorization to become authorized representative,importer or distributor
Suggestion: obtain letter of authorization from relevant party to become
authorized representative, importer or distributor.
Establish and maintain written agreement with relevant party
Written agreement shall be part of the GDPMD system (distribution agreement,
regulatory agreement, etc.)
Establish and maintain communication channel and feedback mechanisms with relevant party
Suggestion: create status report providing medical device feedback information
to relevant party (Post-Market Surveillance)
Manage and communicate with users, public and authority on medical devices dealing with
Establish and maintain efficient communication channel with manufacturers
19
Communication channel
20
Receipt of stock
21
Storage and stock handling
22
Stock rotation
23
Delivery to customers
24
Control
of
nonconforming
medical
devices
including
returned
medical
devices
Establish
feedback
mechanism
for
collecting
comments
and
complaints
from
users
and
public,
to
be
Suggestion:
create
complaint
form
(eg:
8D
report)
forwarded
to
relevant
party
as
applicable
Establish
mechanism
to
provide
information
on
maintenance
services,
including
calibration,
provision
of
spare
suggestion:
include
in
Manuals,
IFU,
leaflets,etc.
parts
and
other
services,
to
the
users
Establish
and
implement
inspection/other
activities
to
ensure
medical
device
meets
specified
requirements Suggestion:
Perform
incoming
inspection,
create
incoming
inspection
log
Maintain
records
of
verification
Inspection
records
shall
be
part
of
the
GDPMD
system
Identify
storage
measures
for
specific
medical
devices
and
stored
in
accordance
with
manufacturer's
Suggestion:
Establish
procedure/work
instruction
for
storage
and
stock
handling
instructions
Provide
suitable
storage
Provide
suitable
storage
area
eg.
Warehouse
Maintain
updated
distribution
records
of
medical
devices
dealing
with
Maintain
distribution
records
of
medical
devices
it
deals
with,including
the
make,
model,
batch
number,
serial
number
and
quantity.
Establish
adequate
precautions
and
control
to
prevent
deterioation
or
damage
of
medical
devices
Can
be
part
of
the
procedure/work
instruction
for
storage
and
stock
handling
Quarantine
status
and
quarantine
area
Provide
proper
identification
and
quarantine
area/storage
for
nonconform
products
Any
system
replacing
physical
quarantine
should
provide
equivalent
security
Storage
for
medical
device
presenting
special
risks
of
abuse,
fire
or
explosion
Provide
proper
storage
area
for
medical
device
presenting
special
risks
of
abuse,
fire
or
explosion
Identification
and
withdrawn
of
broken/damaged
medical
device
from
usable
stock
Provide
proper
identification
for
broken/damaged
medical
device
and
make
sure
the
medical
devices
are
withdrawn
from
usable
stock
(eg.
Store
in
quarantine
area)
Establish
stock
rotation
system
eg.
First-In-First-Out
(FIFO)
system
Identification
of
expired
medical
device
Provide
proper
identification
and
storage
area
for
medical
device
with
a
shelf-life
Dispose
expired
medical
device
Verify
that
certificate
of
registration,
license
and
other
applicable
documents
and
instructions
for
use
(IFU)
is
accompanied
with
registered
medical
device
Ensure
that
medical
device
bears
all
information
needed
Ensure
medical
device
should
only
be
sold/distributed
by
designated
persons
or
entities
only
Provide
proper
documentation
to
customers
Keep
record
of
delivery
transaction
Obtain
condition
of
storage,
transportation,
installation,
testing
and
commissioning
requirements,
users
and
service
manual,spare
part
list
and
certificate
from
manufacturer
and
provide
to
customer
Ensure
delivery
of
medical
device
adhere
specified
condition
Establish
specialized
method
of
delivery
from
the
point
of
collection
to
the
point
of
delivery
Ensure
delivery
of
medical
devices
presenting
special
risks
of
abuse,
fire
or
explosion
are
stored
in
safe,
and
transported
in
safe
conditions
Establish
documented
procedure
Refer to section Disposal of medical devices (clause 25)
Suggestion: create procedure/work instruction for delivery to customer
check whether all informations are available
suggestion: create list of approved distributor/customer
eg. : manuals, delivery notes with traceability information, etc
Records of delivery (eg. Delivery notes) shall be maintained
ensure all informations are obtained from manufacturer and provide to customer
eg.temperature control, special storage condition, etc.
suggestin: establish contract with forwarders
eg. Fire proof cabinet, etc.
Create control of nonconforming medical device procedure
Ensure
nonconforming
medical
device
is
identified
and
controlled
to
prevent
its
unintended
delivery
and
use To
be
included
in
the
procedure
Define
control
and
responsibilities
and
authorities
for
nonconforming
medical
device
handling
Deal
with
nonconforming
medical
device
with
one
or
more
of
following
ways:
-
by
taking
action
to
eliminate
detected
nonconformity
-
by
authorizing
delivery
and
use
under
concession
Ensure
nonconforming
medical
device
is
delivered
and
used
by
concession
only
if
regulatory
requirements
are
met
Maintain
records
of
justification
and
identify
person
authorizing
the
concession
25
26
27
28
29
Disposal of medical devices
Traceability
Maintain records
To be included in the procedure and form. The person authorizing concession
should be identified.
Records shall be part of GDPMD system
Take appropriate action when nonconforming medical device is detected after delivery
Implement Field Corrective Action if required. Refer to provision 40
Establish documented procedure
Create disposal of medical device procedure explaining how devices shall be
disposed (considering locl regulations).
Identify storage area (eg. Quarantine area)
Ensure,
if
the
medical
device
have
not
been
immediately
sent
for
disposal,
they
shall
be
kept
in
clearly
segregated,
safe
and
secured
area
and
identified
Maintain
records
Maintain
updated
records
providing
traceability
of
medical
devices
Records
retention
Ensure all parties involved in the supply chain shall be identifiable
Establish measures to ensure traceability of medical devices throughout distribution channels from
manufacturer/importer to the customer and to the patient
Records including expiry dates and batch records
Specific traceability
Establish tracking records for all implants
requirements for implantable If tracking is not possible for any individual medical devices, the tracking system is still required as follows:
medical devices
(i) to track the medical devices down to healthcare facility level
(ii) to keep track of the following:
- date of medical device was put into service/implanted into a patient
- date the device permanently retired from use or for an implanted medical device, the date it was implanted
Submit surveillance reports to authority at least once a year
Specific requirements for active Establish documented procedure and work instructions for performing installation, testing and
medical devices
commissioning and maintenance activities
Establish and maintain documented procedures, work environments and reference materials, tools and test
equipment and reference measurement procedures, for performing servicing activities including calibration,
repair, maintenance and verifying that they meet the regulatory requirements and applicable standards.
Outsourced activities
Establish
documented
requirements
which
contain
acceptance
criteria
for
installation,
testing
and
commissioning
of
medical
device.
Establish
installation
qualification
and
maintain
adequate
installation
and
inspection
instructions
for
medical
device
requiring
specified
installation
requirements,
and
where
appropriate,
test
procedure
Ensure
proper
installation,
testing
and
commissioning
Ensure
equipment
used
for
testing,
maintenance
and
conservation
of
medical
devices
are
calibrated
or
verified
at
specific
intervals
Ensure
calibration
and
maintenance
of
test
equipments
conforms
to
applicable
standards
maintain
testing
and
commissioning,
installation,
calibration
and
maintenance
service
records.
Ensure
control
outsourced
process
within
GDPMD
scope
Establish
requirements
to
ensure
outsourced
activities
conform
specified
requirements
Ensure
type
and
extent
of
control
applied
to
supplier
are
dependent
on
impact
on
meeting
the
requirements
of
GDPMD
ensure
outsourced
supplier
is
audited
as
part
of
establishment
unless
the
supplier
is
already
certified
to
GDPMD
covering
the
scope
of
outsourced
activities
develop
written
agreements
with
outsourced
party
Physically
segregated
from
other
medical
devices
to
avoid
any
confusion
Records of disposal shall be part of GDPMD system
Maintain a list for customer/serial number of lot number
specify records retention period
- as specified in regulatory requirements, at least equivalent to lifetime of
medical device as defined by manufacturer of medical device,or no less than two
years from the date that medical device is shipped from establishment,
whichever is the longest.
identify the distributor, importer, manufacturer, authorized representative, etc.
create traceability record
Maintain a record for batch number/expiry date
create implant tracking record/log to include all informations needed
submit yearly report to MDA
create installation, testing and commissioning and maintenance procedure and
work instructions (eg: MRI, XRay,etc..)
perfom installation qualification following the manufacturer protocol.
specify calibration or verification interval in work instruction
records should be part of GDPMD system
outsourced activities: eg. warehouse, sterilization, logistic, etc.
All outsouced activities should be closely monitored (auditing for example)
ensure outsourced supplier is audited
create agreement format and maintain records of agreement
identify segregation area
30
Counterfeit
adulterate,
unwholesome
and
tampered
medical
devices
31
Secondary
assembly
including
Availability
of
information
that
describes
characteristics
of
medical
devices
Ensure
all
informations
(instruction
for
use,
maintenance
activities,
etc.)
are
repackaging
available
Availability
of
documented
procedures,
documented
requirements,
work
instructions
and
reference
materials
Ensure
all
documents
are
available
during
secondary
assembly
activities
and
reference
measurement
procedures
as
necessary
The
use
of
suitable
equipment
packaging
machine,
label
printer,
etc.
Availability
and
use
of
monitoring
and
measuring
devices
ensure
suitable
monitoring
and
measuring
device
are
used
Implementation
of
monitoring
and
measurement
activities
first
piece
inspection,
statistical
process
control
(SPC),
etc.
Implementation
of
release
of
medical
device,
their
delivery
and
post-delivery
activities
final
release
inspection,etc.
Implementation
of
defined
operations
and
packaging
of
medical
devices
ensure
packaging
activities
are
according
to
packaging
work
instruction
Establish
and
maintain
a
record
for
each
batch
of
medical
devices
that
provides
traceabilty
and
identifies
the
eg.
Work
order,
tracking
records
amount
assembled
and
the
amount
approved
for
distribution
Ensure
batch
record
shall
be
verified
and
approved
by
qualified
personnel
suggestion:
include
responsibility
of
verification
of
batch
record
in
final
release
procedure
Assembly
documents
Kept
batch
assembly
records
maintain
assembly
records
eg.work
order,
operator
tracking
record,
etc.
Ensure
assembly
are
made
or
completed
at
each
time
action
is
taken
to
trace
significant
activities
of
assembly eg.
Verification
by
supervisor
is
needed
after
each
assembly
activities
completed
32
inform the Authority and manufacturer immediately.
Record retention
33
Materials control
inform authority through http://www.mdb.gov.my
as specified in regulatory requirements, at least equivalent to lifetime of medical
device as defined by product owner of medical device,or no less than two years
from the date that medical device is shipped from establishment, whichever is
the longest.
For each delivery, incoming medical device are checked for integrity of package and seal, for correspondence perform incoming inspection, label verification, etc.
between delivery note and the supplier's label, and for compliance with quality specification
medical device with breached primary package are not used for secondary assembly
locate in proper segregation area
medical device in storage area are appropriately labeled
Identify properly (labelling, storage area, etc.)
33
Materials control
34
Labelling
35
Good assembly practices
36
38
39
40
41
42
43
44
45
Appropriate
procedures
or
measures
are
taken
to
assure
identity
of
the
contents
of
each
packing
of
medical
device
Clearly
identify
bulk
containers
from
which
quantities
of
medical
device
that
have
been
drawn
Placed
medical
device
requiring
special
storage
conditions
in
areas
which
are
designed
an
equipped
to
provide
desired
conditions
Continuously
monitor
and
record
storage
and
conditions
Express
actual
storage
temperature
quantitatively
the
purchase,
handling,
and
control
of
all
packaging
materials
are
accorded
attention
similar
to
that
given
to
starting
materials
packaging
materials
are
issued
for
use
only
by
authorized
personnel
in
accordance
with
documented
procedure
when
setting
up
a
program
for
packaging
operations,
particular
attention
is
given
to
minimize
risk
if
cross-
contamination,
mix-ups
or
substitutions
different
medical
devices
shall
not
be
packaged
in
close
proximity
unless
there
is
physical
segregation
Ensure
repackaged
medical
devices
bear
all
original
labelling
(including
IFU
and
any
other
information
sheet
or
leaflet,
etc.)
and
all
labelling
information,
except
for
quantity
and
distributor
identity.
Check
medical
devices
and
materials
used
for
assembly
before
use
by
a
designated
person
for
quantity,
identity
and
conformity
with
the
packaging
instructions.
Performed
line
clearance
prior
commencement
of
assembly
operation
Check
and
record
the
correct
performance
of
any
printing
operation
which
is
carrried
out
separately
or
in
the
course
of
packaging
Re-check
printing
by
hand
at
regular
intervals
Assembly
equipment/apparatus
are
cleaned
according
to
detailed
and
written
procedures
and
stored
only
in
a
clean
and
dry
condition
Assembly
equipment/apparatus
do
not
present
any
hazard
to
the
medical
devices
Parts
of
assembly
equipment/apparatus
that
come
into
contact
with
medical
devices
do
not
effect
quality
of
medical
devices
and
present
any
hazard
Calibrate
and
check
control
environment
at
defined
intervals
and
adequate
records
of
calibration
shall
be
maintained.
Create procedure/work instruction
Conduct internal audits at planned intervals
Maintain records of audit and results
Take action to eliminate detected nonconformities and causes without undue delay
Establish documented procedure
Establish documented procedure
Internal audit plan should be created
Records of audit shall be part of GDPMD system
Action should be taken immediately
Create corrective action procedure and form.
Create preventive action procedure and form.
Suggestion: corrective action and preventive action procedure can be combined
in one procedure
create proper labeling/identification/storage area
eg. Medical device that needs temperature control
create storage and condition monitoring record
record storage temperature in storage and condition monitoring record
ensure material control activities are according to procedure/work instruction
include responsibility of issuing packaging material in procedure/work instruction
proper identification, segregation area
create proper segregation area
perform labelling inspection/verification
perform inspection. Medical device and material used should be inspected by
designated personnel only.
suggestion: include line clearance activity in work instruction
perform label verification
perform cleaning according to cleaning procedure
calibration interval and checking of control environment should be specified in
procedure or etc.
records should be maintained
Quality control
Finished medical device assessment shall embrace all relevant factors,including assembly conditions, a review create final release procedure, inspection criteria, etc.
of packaging documentation, compliance with finished medical device specification and visual examination of Perform inspection/verification according to specified requirement
final finished pack
Process of secondary assembly shall not compromise conformity of medical device to essential principles of
safety and performance
Medical device complaints
Establish documented procedure
Create complaint handling procedure
Review all complaints and other information concerning potentially defective and counterfeit medical
To be included in complaint procedure and form (review section)
devices, including description of action to be taken and reporting to relevant parties, where appropriate
Record any complaint and investigate to identify origin or the reason for complaint
Create complaint form and complaint log
Maintain records of complaints, investigation and action taken
Create complaint form
Follow-up action should be taken after investigation and evaluation of complaint, where necessary
To be included in complaint form
Distribution records
Document all activities relating to distribution
Maintain distribution records including receipts, storage, delivery and disposal
FCA and FSN
Establish documented procedure
Create procedure for handling of FCA and FSN
Define responsibilities for planning, conducting and reporting of corrective actions in procedure
Include responsibilities in procedure
Establish writing a recall/withdrawal procedure in consultation with manufacturer
Establish recall procedure
Inform authority prior execution of FCA and FSN
Any adverse event shall be reported through mdb website:
http://www.mdb.gov.my/mdb/index.php?option=com_content&task=view&id=
19&Itemid=115
Inform customers to whom medical device was distributed with appropriate degree of urgency
customer should be informed immediately
Inform overseas counterparts on FCA and FSN if medical devices are exported
Communicate with overseas counterpart if medical devices are exported
Maintain records of action taken and approval by manufacturer and authority
Records shall be maintained
Recall
Establish documented procedure
Recommendation: Recall can be included in FCA procedure
Ensure the system comply with regulatory requirements
Manufacturer and/or authorized representative shall be informed in the event of recall
Inform Manufacturer and/or authorized representative
where recall is instituted by an entity other than manufacturer and/or authorized representative,
consult Manufacturer and/or authorized representative if recall is instituted by
consultation with manufacturer and/or authorized representative should, where possible, take place before other entity
recall is instituted
recall information shall be reported to the Authority
inform authority through http://www.mdb.gov.my
Progress of recall process should be recorded and a final report issued, which includes a reconcilliation
issue final report of recall process
between delivered and recovered quantities of products.
Mandatory problem reporting Establish documented procedure
Establish mandatory problem reporting procedure
Each incident report shall lead to a final report where corrective actions are applicable
create mandatory problem report form
Internal audits
Establish documented procedure
Define responsibilities,requirements for planning and conducting audits and
reporting of result and maintenance of audit records.
Plan an audit program
Establish audit plan
Define audit criteria, scope, frequency and methods
audit criteria, scope, frequency and methods should be included in the procedure
Corrective
action
Preventive
action
También podría gustarte
- Sample Management Review Agenda and MinutesDocumento6 páginasSample Management Review Agenda and MinutesTravis100% (3)
- ISO 13485 Internal Audit ChecklistDocumento23 páginasISO 13485 Internal Audit Checklistenrique59197291% (23)
- Quality ManualDocumento19 páginasQuality ManualKung Karona100% (2)
- MDPWG Ahmad Shariff Hambali Good Distribution For Medical Device GDPMDDocumento40 páginasMDPWG Ahmad Shariff Hambali Good Distribution For Medical Device GDPMDnaravichandran3662Aún no hay calificaciones
- SOP of Release or Rejection of Finished ProductDocumento1 páginaSOP of Release or Rejection of Finished ProductPrince MoniAún no hay calificaciones
- SOP On Monitoring of Temperature and Relative Humidity.Documento2 páginasSOP On Monitoring of Temperature and Relative Humidity.RainMan75100% (2)
- SOPSP05 VendorSelection BSDocumento3 páginasSOPSP05 VendorSelection BStroubledsoul100% (1)
- Audit Checklist For ISO 13485Documento6 páginasAudit Checklist For ISO 13485EdAún no hay calificaciones
- Capa FormDocumento3 páginasCapa FormZahid Nazir100% (1)
- Achieving and Maintaining Qualified Facilities with a Validation Master Plan (VMPDocumento3 páginasAchieving and Maintaining Qualified Facilities with a Validation Master Plan (VMPPrince MoniAún no hay calificaciones
- 7 steps CAPA medical devicesDocumento7 páginas7 steps CAPA medical devicesShantha Kumar ParameswaranAún no hay calificaciones
- Product Recall and Withdrawal ProcedureDocumento5 páginasProduct Recall and Withdrawal ProcedureNaizil Kareem100% (5)
- Quality System Manual: ISO 13485 U.S. QSR (21 CFR 820)Documento27 páginasQuality System Manual: ISO 13485 U.S. QSR (21 CFR 820)Khalid Mehboob100% (1)
- FDA Medical Device Complaint FormDocumento2 páginasFDA Medical Device Complaint Formclayton51175% (4)
- 93-42-EC ER Checklist ExampleDocumento16 páginas93-42-EC ER Checklist Exampledulichsinhthai100% (2)
- 410 09e Checkliste For Assessment 13485 MDDDocumento51 páginas410 09e Checkliste For Assessment 13485 MDDeko Setyawan100% (1)
- ISO 13485 and ISO 9001 Correspondence GuideDocumento4 páginasISO 13485 and ISO 9001 Correspondence GuideGadus Spp100% (4)
- Quality Manual ExampleDocumento32 páginasQuality Manual ExamplesaiaviAún no hay calificaciones
- 820 - ISO 13485-2003 Comparison MatrixDocumento37 páginas820 - ISO 13485-2003 Comparison MatrixAli Imamudeen100% (2)
- GMP Audit Checklist for Pharmaceutical ManufacturingDocumento31 páginasGMP Audit Checklist for Pharmaceutical ManufacturingSandra100% (1)
- MRF Management ReviewDocumento4 páginasMRF Management ReviewhastinkakaAún no hay calificaciones
- ISO 13485 QMS manual indexDocumento5 páginasISO 13485 QMS manual indexQuality and Safety Consultants Co.Aún no hay calificaciones
- SOP For Internal AuditDocumento3 páginasSOP For Internal AuditMuhammad Nisar75% (4)
- Calibration Policy SOPDocumento3 páginasCalibration Policy SOPYousifAún no hay calificaciones
- 21 CFR 820 Audit ChecklistDocumento7 páginas21 CFR 820 Audit ChecklistFrankMartinez100% (1)
- ISO 13485 Gantt Chart - May 2020Documento4 páginasISO 13485 Gantt Chart - May 2020Rohini Gadhawe100% (4)
- 2016 StandardDocumento39 páginas2016 StandardAnkur Dhir100% (4)
- Calibration and Maintenance of Measuring and Monitoring Devices SOPDocumento3 páginasCalibration and Maintenance of Measuring and Monitoring Devices SOPBPG Service100% (1)
- Internal Quality Audit Checklist ISO 13485:2003 & ISO 9001:2000Documento29 páginasInternal Quality Audit Checklist ISO 13485:2003 & ISO 9001:2000JocelyneAún no hay calificaciones
- Technical FilesDocumento15 páginasTechnical Fileshitham shehataAún no hay calificaciones
- Managemnet Review SOP & RecordsDocumento3 páginasManagemnet Review SOP & RecordsPritam MitraAún no hay calificaciones
- OOS FormatDocumento4 páginasOOS Formatbpharmba100% (1)
- Pecb Iso 13485 Lead Auditor Exam Preparation GuideDocumento15 páginasPecb Iso 13485 Lead Auditor Exam Preparation GuiderberrospiAún no hay calificaciones
- 2016 Ia Sample ChecklistDocumento5 páginas2016 Ia Sample ChecklistMiss BreedloveAún no hay calificaciones
- MDR Readiness Checklist: Prepared by Cite Medical SolutionsDocumento37 páginasMDR Readiness Checklist: Prepared by Cite Medical SolutionsBeal100% (1)
- Good Distribution Practice For Medical Device108201532845PM1Documento60 páginasGood Distribution Practice For Medical Device108201532845PM1Sean100% (1)
- QSP-001633 - Rev 04 - Luminus Testing Laboratory Quality Manual207Documento34 páginasQSP-001633 - Rev 04 - Luminus Testing Laboratory Quality Manual207SureshAún no hay calificaciones
- ISO 13485 Process MatrixDocumento2 páginasISO 13485 Process MatrixManjunath BAún no hay calificaciones
- ISO 13485 PurchasingDocumento5 páginasISO 13485 PurchasingSubhashAún no hay calificaciones
- ISO 13485 Audit Report for Haiyan Kangyuan MedicalDocumento30 páginasISO 13485 Audit Report for Haiyan Kangyuan MedicalAymen ZebbarAún no hay calificaciones
- Iso 13485 Manual DocumentsDocumento14 páginasIso 13485 Manual DocumentsRRR1100% (1)
- ISO 13485 ComparisonDocumento6 páginasISO 13485 ComparisonMikeAún no hay calificaciones
- Audit TcoDocumento21 páginasAudit Tcoali reza malaekehAún no hay calificaciones
- Audit Checklist - IsO 13485 and MDDDocumento6 páginasAudit Checklist - IsO 13485 and MDDMarcos Poffo100% (2)
- MammographyDocumento27 páginasMammographyRohmatullah Anas100% (1)
- Standard Awareness Training 13485Documento21 páginasStandard Awareness Training 13485Jeelani BashaAún no hay calificaciones
- Cracking the Case of ISO 9001:2015 for Service: A Simple Guide to Implementing Quality Management in Service OrganizationsDe EverandCracking the Case of ISO 9001:2015 for Service: A Simple Guide to Implementing Quality Management in Service OrganizationsAún no hay calificaciones
- Preparing For Certification AuditDocumento58 páginasPreparing For Certification AuditAntonio P. SantosAún no hay calificaciones
- A Sample of The Completed Essential Principles Conformity Checklist MD CCLDocumento12 páginasA Sample of The Completed Essential Principles Conformity Checklist MD CCLAyman Ali100% (1)
- The ASQ Certified Quality Auditor HandbookDe EverandThe ASQ Certified Quality Auditor HandbookCalificación: 1 de 5 estrellas1/5 (2)
- ISO 13485 Lead Auditor Two Page BrochureDocumento2 páginasISO 13485 Lead Auditor Two Page BrochurePECBCERTIFICATIONAún no hay calificaciones
- Silabus Lead Auditor 9001Documento4 páginasSilabus Lead Auditor 9001Bobby Febri KrisdiyantoAún no hay calificaciones
- CLAUSE 8.5 Production and Service ProvisionDocumento10 páginasCLAUSE 8.5 Production and Service ProvisionNavnath TamhaneAún no hay calificaciones
- Guidance On The Rules of Classification For General Medical DevicesDocumento29 páginasGuidance On The Rules of Classification For General Medical Devicesnaravichandran3662Aún no hay calificaciones
- GMPDocumento5 páginasGMPharwinderpanditAún no hay calificaciones
- HIMSS EMRAM Stage 7 DMTDocumento20 páginasHIMSS EMRAM Stage 7 DMTclrhoadesAún no hay calificaciones
- MDSAP QMS F0008.2.003 Internal Assessment Checklist 2017-03-01Documento15 páginasMDSAP QMS F0008.2.003 Internal Assessment Checklist 2017-03-01nsk79in@gmail.comAún no hay calificaciones
- Emergency Preparedness Concepts Test BankDocumento13 páginasEmergency Preparedness Concepts Test BankTyson Easo JonesAún no hay calificaciones
- Comprehensive Physical AssessmentDocumento2 páginasComprehensive Physical AssessmentJan Christian Dayto79% (33)
- Facilities and Environmental Conditions (Linked To Vol. III, Sec. 2) PDFDocumento8 páginasFacilities and Environmental Conditions (Linked To Vol. III, Sec. 2) PDFimran shaukatAún no hay calificaciones
- As ISO 14534-2003 Opthalmic Optics - Contact Lenses and Contact Lens Care Products - Fundamental RequirementsDocumento8 páginasAs ISO 14534-2003 Opthalmic Optics - Contact Lenses and Contact Lens Care Products - Fundamental RequirementsSAI Global - APACAún no hay calificaciones
- Control of Nonconforming ProductsDocumento2 páginasControl of Nonconforming ProductsPrince Moni100% (1)
- LS QA ManualDocumento55 páginasLS QA ManualjtwaniAún no hay calificaciones
- Iso 22001 Efresh IndiaDocumento13 páginasIso 22001 Efresh IndiaMahmoud KhalifaAún no hay calificaciones
- F001 GDPMDDocumento5 páginasF001 GDPMDnaravichandran3662Aún no hay calificaciones
- GD0009 Classification of General Medical DeviceDocumento39 páginasGD0009 Classification of General Medical DeviceAdel AdielaAún no hay calificaciones
- DOLE OHSC Audit Report FA1 2018Documento13 páginasDOLE OHSC Audit Report FA1 2018naravichandran3662Aún no hay calificaciones
- Ivd Medical Device Classification SystemDocumento19 páginasIvd Medical Device Classification SystemoytAún no hay calificaciones
- Module 3 - Approaches To Clean Vehicle ProcurementDocumento27 páginasModule 3 - Approaches To Clean Vehicle Procurementnaravichandran3662Aún no hay calificaciones
- How To Apply For In-Vitro Diagnostic (IVD) Medical Device Registration Under Medical Device Act 2012 (Act 737)Documento13 páginasHow To Apply For In-Vitro Diagnostic (IVD) Medical Device Registration Under Medical Device Act 2012 (Act 737)naravichandran3662Aún no hay calificaciones
- Additional Transition Period For Medical Device LabelingDocumento3 páginasAdditional Transition Period For Medical Device Labelingnaravichandran3662Aún no hay calificaciones
- Exemption From Registration Requirement For Export Only Medical Device (Revision 1) BMDocumento3 páginasExemption From Registration Requirement For Export Only Medical Device (Revision 1) BMnaravichandran3662Aún no hay calificaciones
- How To Apply For Establishment License Under Medical Device Act 2012 (Act 737) V2 MalaysiaDocumento7 páginasHow To Apply For Establishment License Under Medical Device Act 2012 (Act 737) V2 Malaysianaravichandran3662Aún no hay calificaciones
- E-MDES Manual 2020Documento152 páginasE-MDES Manual 2020naravichandran3662Aún no hay calificaciones
- MDA Establishment Licence RenewalDocumento14 páginasMDA Establishment Licence Renewalnaravichandran3662Aún no hay calificaciones
- Post-Market Responsibilities For Manufacturer and Authorized Representative (Revision 1)Documento3 páginasPost-Market Responsibilities For Manufacturer and Authorized Representative (Revision 1)naravichandran3662Aún no hay calificaciones
- 771 1Documento25 páginas771 1yogiforyouAún no hay calificaciones
- Medical Device (Declaration) Order 2017Documento5 páginasMedical Device (Declaration) Order 2017naravichandran3662Aún no hay calificaciones
- MDARR No 1 First Revision November 15Documento26 páginasMDARR No 1 First Revision November 15popopioAún no hay calificaciones
- Selangor - Senarai Klinik Perubatan Swasta Berdaftar 2018 (Update 260319)Documento115 páginasSelangor - Senarai Klinik Perubatan Swasta Berdaftar 2018 (Update 260319)naravichandran3662Aún no hay calificaciones
- Barriers To CommunicationDocumento34 páginasBarriers To Communicationnaravichandran3662Aún no hay calificaciones
- Compilation of Guidelines For Redress of Public Grievances - Naresh KadyanDocumento290 páginasCompilation of Guidelines For Redress of Public Grievances - Naresh KadyanNaresh KadyanAún no hay calificaciones
- Medical Device ACT 2012 (Act 737)Documento0 páginasMedical Device ACT 2012 (Act 737)Chee Chiow NeoAún no hay calificaciones
- Medical Device Authority Act 2012 (Eng) PDFDocumento24 páginasMedical Device Authority Act 2012 (Eng) PDFnaravichandran3662Aún no hay calificaciones
- SENARAI HOSPITAL SWASTA BERLESEN DI JOHOR DAN NEGERI-NEGERI LAINDocumento23 páginasSENARAI HOSPITAL SWASTA BERLESEN DI JOHOR DAN NEGERI-NEGERI LAINnaravichandran3662Aún no hay calificaciones
- WPKL Senarai Klinik Perubatan Swasta Berdaftar 2018 (Update 260319)Documento65 páginasWPKL Senarai Klinik Perubatan Swasta Berdaftar 2018 (Update 260319)naravichandran3662Aún no hay calificaciones
- Barriers To CommunicationDocumento34 páginasBarriers To Communicationnaravichandran3662Aún no hay calificaciones
- Quality and Costs of Primary Care - Malaysia Phase 1 ReportDocumento170 páginasQuality and Costs of Primary Care - Malaysia Phase 1 Reportnaravichandran3662Aún no hay calificaciones
- MDA - GD0026 - Requirement On Labelling of MD 3rd EdDocumento13 páginasMDA - GD0026 - Requirement On Labelling of MD 3rd Ednaravichandran3662Aún no hay calificaciones
- E-Book Ophthalmology Referral Guide For GPsDocumento11 páginasE-Book Ophthalmology Referral Guide For GPsnaravichandran3662Aún no hay calificaciones
- Are Malaysian Police Really StressedDocumento10 páginasAre Malaysian Police Really Stressednaravichandran3662Aún no hay calificaciones
- Peaceful Assembly Act 736Documento21 páginasPeaceful Assembly Act 736William AnthonyAún no hay calificaciones
- Peaceful Assembly Act 736Documento21 páginasPeaceful Assembly Act 736William AnthonyAún no hay calificaciones
- DR Wong Teck WeeipadDocumento2 páginasDR Wong Teck Weeipadtwwong68Aún no hay calificaciones
- Philips Respironics Bipap ST Niv Noninvasive VentilatorDocumento2 páginasPhilips Respironics Bipap ST Niv Noninvasive Ventilatorsonia87Aún no hay calificaciones
- Fa14 Concept Map 10-26-14 2Documento2 páginasFa14 Concept Map 10-26-14 2api-270415831Aún no hay calificaciones
- Nextier - Pharma 101 - 29-Jun-2016Documento28 páginasNextier - Pharma 101 - 29-Jun-2016Ammar ImtiazAún no hay calificaciones
- Apotex Pharmachem Product ListDocumento2 páginasApotex Pharmachem Product Listমোঃ এমদাদুল হকAún no hay calificaciones
- List of Manufacturing Units With Valid Ayush 64 Technology Transfer Agreement As On 15.06.2021Documento5 páginasList of Manufacturing Units With Valid Ayush 64 Technology Transfer Agreement As On 15.06.2021Sunira EnterprisesAún no hay calificaciones
- Hesi v2Documento125 páginasHesi v2kandiezein88Aún no hay calificaciones
- Evaluation of scales in immune-mediated polyneuropathiesDocumento222 páginasEvaluation of scales in immune-mediated polyneuropathiesAngelo MalerbaAún no hay calificaciones
- Hand Hygiene: Protocol For UWS ClinicsDocumento8 páginasHand Hygiene: Protocol For UWS ClinicsOliaAún no hay calificaciones
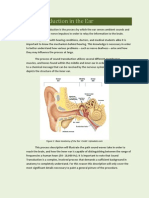
- Sound Transduction EarDocumento7 páginasSound Transduction Earhsc5013100% (1)
- Unit-V Chapter 20. Breathing and Exchange of Gases: Important PointsDocumento13 páginasUnit-V Chapter 20. Breathing and Exchange of Gases: Important PointsminaAún no hay calificaciones
- Reflex ActionDocumento26 páginasReflex ActionMudassar RoomiAún no hay calificaciones
- Jurnal InvaginasiDocumento3 páginasJurnal InvaginasiAbdurrohman IzzuddinAún no hay calificaciones
- A Review On Acacia Arabica - An Indian Medicinal Plant: IJPSR (2012), Vol. 3, Issue 07 (Review Article)Documento11 páginasA Review On Acacia Arabica - An Indian Medicinal Plant: IJPSR (2012), Vol. 3, Issue 07 (Review Article)amit chavanAún no hay calificaciones
- Depression in Today - BioDocumento5 páginasDepression in Today - Bioapi-272648963Aún no hay calificaciones
- Sage Char-Lee ResumeDocumento2 páginasSage Char-Lee Resumeapi-510197139Aún no hay calificaciones
- (Schedule P: Drugs and Cosmetics Rules 1945Documento7 páginas(Schedule P: Drugs and Cosmetics Rules 1945rpadmakrishnanAún no hay calificaciones
- Clinical Experience With The Constellation Vision SystemDocumento16 páginasClinical Experience With The Constellation Vision SystemMohammad Abdullah BawtagAún no hay calificaciones
- FISIOGRAFT ENG Set 17 PDFDocumento28 páginasFISIOGRAFT ENG Set 17 PDFmaxAún no hay calificaciones
- Hypertension: Hozan Jaza MSC Clinical Pharmacy College of Pharmacy 10/12/2020Documento81 páginasHypertension: Hozan Jaza MSC Clinical Pharmacy College of Pharmacy 10/12/2020Alan K MhamadAún no hay calificaciones
- Bonifacio V. Romero High School Tle Beauty Care 9 1st Quarter Examination S.Y. 2018-2019Documento6 páginasBonifacio V. Romero High School Tle Beauty Care 9 1st Quarter Examination S.Y. 2018-2019Virginia Saavedra100% (1)
- Masterlist SummaryDocumento6 páginasMasterlist Summarykentclark03Aún no hay calificaciones
- Staph and Strep Lab Report BMS 501Documento2 páginasStaph and Strep Lab Report BMS 501aruiz93Aún no hay calificaciones
- NephrologyDocumento5 páginasNephrologyPooja NalluriAún no hay calificaciones
- DR Koenig Redacted Expert Report Publicly FiledDocumento14 páginasDR Koenig Redacted Expert Report Publicly FiledBrit StackAún no hay calificaciones
- Biology Contents - RPSC FSO by Food TecKnowDocumento35 páginasBiology Contents - RPSC FSO by Food TecKnowRahul JainAún no hay calificaciones